FACT 1 Study Information Sheet
Dear Colleagues,
Thank you for your recent expression of interest in partaking in the first Foot & Ankle Collaborative in Trauma (FACT) study which is looking at Talus fracture and is led by Mr Ed Gee. This study and subsequent FACT studies are badged by BOFAS.
Background
Study Protocol
Full details and the study protocol can be viewed here: https://www.darfi.net/studies/fact-1/ where you can review and download the latest version of the protocol directly.
Study Inclusion/Exclusion Criteria
Inclusion Criteria
- Adult aged 16 or above at date of injury
- Injury sustained between 1st January 2013 and 28th February 2026
- Minimum clinical and radiographic follow up of 3 months
Exclusion Criteria
- Children under the age of 16 at date of injury
- Incomplete follow up less than 3 months
- Patients that are excluded via the National Data opt-out scheme
Study Timeline
The following is a provisional study timeline for
- Data collection: 15th February 2026 – 12th June 2026
- Data cleaning & quality control: 15th June 2026 – 17th July 2026
- Deadline for response to data queries: 4th August 2026
- Data analysis: August – October 2026
- Initial presentation of results: BOFAS 2026 (4th – 6th November 2026 in Cardiff)
- Write up and publication of results: November 2026 – January 2027
How To Get Involved
- Make sure your Trust is registered to DARFI in order for us to be able to accept your data. You can check if your Trust is on our list of current active and onboarded DARFI sites via this link: https://www.darfi.net/sites/. If it is, contact your local study PI and let them know you are keen to get involved for them to update the delegation log and ensure you are credited for your.
- If your Trust is not on the list above, please complete an Expression of Interest Form (https://www.darfi.net/expression-of-interest/). This will then be picked up by our R&I colleagues who will follow this up and ensure all the relevant agreements are in place between LUHFT and your NHS Trust. Once completed, this will then mean that each registered/onboarded site can partake in this study as well as any future DARFI studies instantly without any further R&I agreements.
- Review the FACT-1 study protocol for all the study specific details
- Please liaise with your audit/research teams, relevant consultants, and business intelligence departments to identify patients for inclusion in the study. To assist with this, you can request searches using the following diagnosis and procedure codes.
- ICD-10 code: S92.1
- Procedure codes (OPCS-4): W20.2 ± Z84.*, W24.2, W22.2, plus ankle equivalents (W20.1, W24.1, W22.1) to catch miscoding
- SNOWMED CT codes: 3381004 / 79626009 / 208684008 / 208685009 / 208686005 / 208696001 / 208697005 / 208698000 / 263246003 / 429664005
- Download the data collection sheet and radiograph PowerPoint template from the downloads section below
- Review the data collection video before you starting your data collection
- Once you’ve completed the data collection, send your completed data collection sheet and radiograph PowerPoint as per the instructions below.
YouTube Video
Rules of Engagement
Two doctors (a senior and a junior PI) will be listed as authors for the first 10 patients contributed by each centre. Thereafter, each centre may add one additional author for every further 10 patients included. For example, a centre contributing 43 patients would be eligible for two authors for the initial 10 patients, plus one additional author for each subsequent block of 10 patients, allowing a maximum of six authors in total for that Trust.
Classifications
Hawkins Classification (For talar neck fractures): The original Hawkins classification includes three fracture types: Type I through Type III. The modern version includes Type IV, which was added by Canale and Kelly in 1978 and described as follows with more details (diagrams/radiographs) available here:
- Type I: Fracture is a talar neck fracture
- Type II: A talar neck fracture with subtalar dislocation
- Type III: Talar neck fracture with subtalar and tibiotalar dislocations
- Type IV: is a talar neck fracture with subtalar, tibiotalar, and talonavicular dislocations.
Sneppen Classification (For talar body fractures): The original Sneppen classification described in 1977 outlines 6 types of talar body fractures
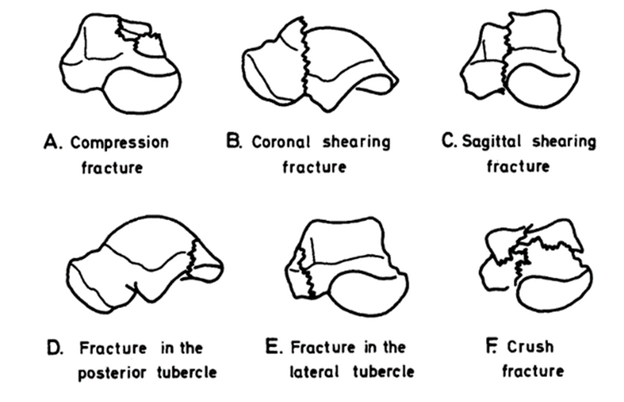
- Type A: Transchondral or Osteochondral (Compression) Fracture: Involves the talar dome, often with compression.
- Type B: Coronal Shear Fracture: A vertical or oblique shear fracture in the coronal plane.
- Type C: Sagittal Shear Fracture: A vertical or oblique shear fracture in the sagittal plane.
- Type D: Posterior Tubercle Fracture: Involves the posterior process (more commonly the lateral tubercle).
- Type E: Lateral Process Fracture: Often termed a “snowboarder’s fracture,” involving the lateral process.
- Type F: Crush Injuries: Comminuted fractures of the talar body, typically from high-energy, axial loading injuries.
Kellgren Lawrence scale: This is a five-grade (0-4) radiographic classification system described in 1957 used to assess the severity of osteoarthritis (OA), primarily in the knee but being repurposed for the talus.
- Grade 0 (None): No radiographic features of OA.
- Grade 1 (Doubtful): Possible, minute osteophytes of doubtful clinical significance; joint space is usually preserved.
- Grade 2 (Mild): Definite osteophytes (bone spurs) with possible joint space narrowing.
- Grade 3 (Moderate): Moderate, multiple osteophytes, definite joint space narrowing, and some sclerosis.
- Grade 4 (Severe): Large osteophytes, marked joint space narrowing, severe sclerosis, and definite deformity of the bone ends.
Upload Your Data
Frequently Asked Questions
Will be updated as they arise.
Contact
For any questions regarding the study, please contact fact1.study@gmail.com which is covered by members of the study steering committee.
PLEASE DO NOT SEND YOUR DATA OR ANY PATIENT INFORMATION TO THAT ADDRESS
To upload your data, please use the button above